|
Deduced levels E, J, π in Rn 2 0 6, 2 0 8. The decay process begins in the nucleus of a neutron-deficient isotope upon the conversion of a proton to a neutron with simultaneous emission of a positron, or +-particle, from the nucleus. Express the changes in the atomic number and mass number of a radioactive nuclei when an alpha, beta, or gamma particle is emitted. The mode of positron decay is particularly advantageous for detection and quantification by external measurement. Compare qualitatively the ionizing and penetration power of alpha particles () ( ), beta particles () ( ), and gamma rays () ( ). RADIOACTIVITY Fr 2 0 6, 2 0 8 from mass separated products of Ir nat ( Ne 20, x n ) Fr 2 0 6, 2 0 8. 17.3: Types of Radioactivity- Alpha, Beta, and Gamma Decay. The energy levels were also compared to the predictions of the interacting boson approximation model. On the other hand, electron capture is always an alternative decay mode for radioactive isotopes with sufficient energy to decay by. This process results in the emission of positron and neutrino. Electron capture is the primary decay mode for isotopes with insufficient energy (Q < 2 x 511 keV) difference between the isotope and its prospective daughter for the nuclide to decay by emitting a positron. Diagram of a Transition That Produces Electron Capture In an electron capture transition, radiation is not emitted directly. The ratio between the two processes is specific for each nuclide. For the positron emission from a nucleus, there is another competing process known as electron capture electron from an inner orbit, say, the K shell. i) Positron: A proton get converted into neutron due to radio-active process. Electron capture often competes with positron emission if a nuclide is a positron emitter, some nuclei will emit positrons and some will capture electrons. 7 4Be + 0 -1e 7 3Li + energy Answer 4: Following are the distinct features of transmutations caused by positron emissions and electron capture. These nuclei appear to be excellent candidates for interpretation in terms of a weak coupling shell model. Following is an example of K-electron capture. ELECTRON CAPTURE: In this process, the inner electron of an atom is consumed by a neutral nucleus. The mass number of the daughter nucleus decreases by 1 in this process. al have calculated the ratio of cross sections for electron capture by positrons to protons in helium form a few to 200 eV and find a curve which. The positron can be thought of as a twin of an electron but with an opposite charge. The energies and many of the spins were determined for 18 excited, even parity states in Rn 208 and for 10 excited, even parity states in Rn 206. In this process, a proton is converted to neutron and positron. The electron-capture and positron decays to Rn 206 and Rn 208 were studied by collecting γ ray and internal conversion electron singles spectra as a function of decay time as well as γ − γ, γ − e −, and γ − x ray coincidence spectra. (a) Electrons are fundamental particles (that is, not made of even smaller particles) that carry one unit of negative charge. When a positron collides with electron annihilation, it results in the production of two or more gamma-ray photons. The mass number is the sum of the numbers of protons and neutrons present. A positron has the equal or same mass as an electron and a spin of 1/2, but it has an electrical charge of +1. Both positron decay and electron capture increase the neutron-to-proton ratio electron capture is more common for heavier elements such those of row 5. 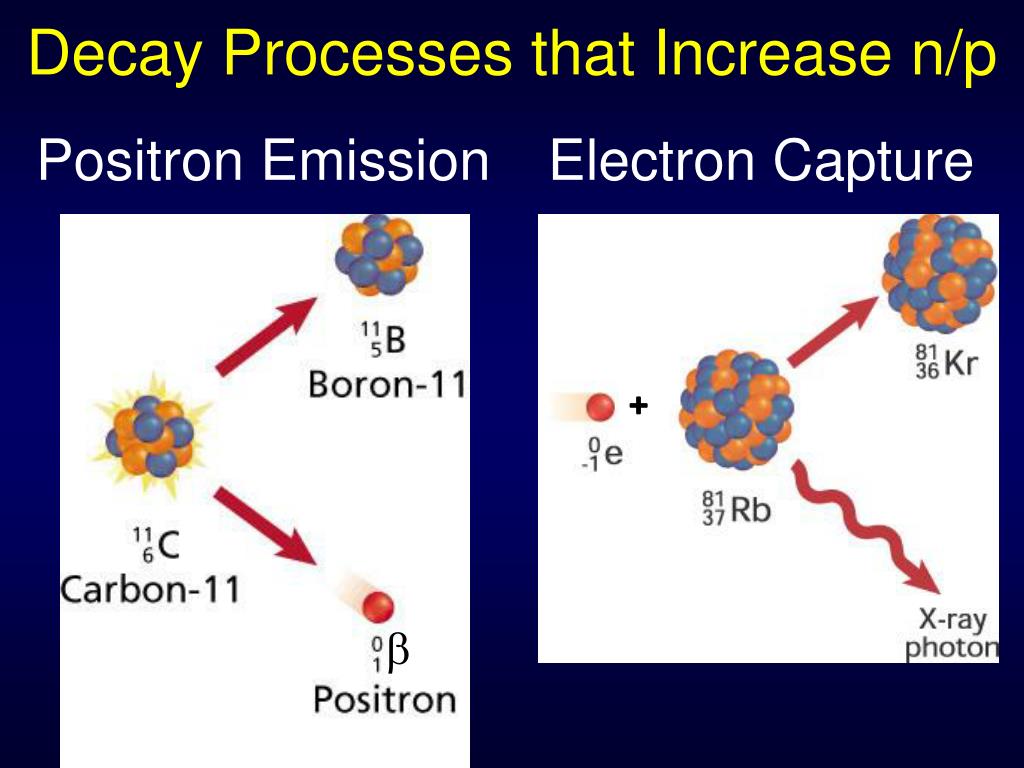
\beta \nonumber\]ī The mass number of the second product is A = 30 − 0 = 30, and its atomic number is Z = 15 − 1 = 14, which corresponds to silicon.The isotopes Fr 206 and Fr 208 were produced by the reactions Ir ( Ne 20, x n ) Fr 2 0 6, 2 0 8 and mass separated on-line. A positron or antielectron is the antimatter counterpart to an electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
